LOCTITE ABLESTIK ABP 2035SCR
- Low stress
- Snap cure
- Good dispense behavior
Product Description
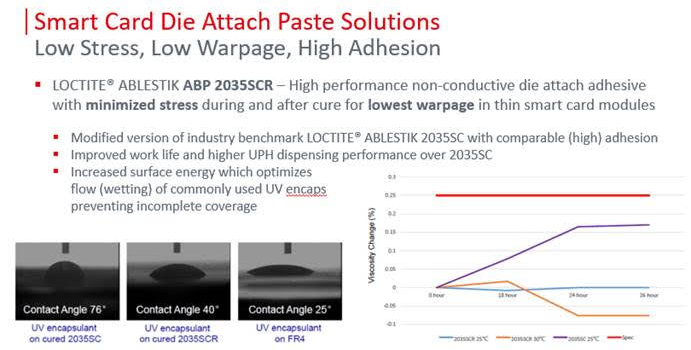
LOCTITE® ABLESTIK ABP 2035SCR non-conductive die attach adhesive has been formulated for use in high throughput die attach applications. This material is a direct replacement for its predecessors LOCTITE ABLESTIK 2033SC and LOCTITE ABLESTIK 2035SC. This BMI/Acrylic material is designed to minimize stress and resulting warpage between dissimilar surfaces. This adhesive has a maximum filler size of less than 25um and an average of less than 15um. Compared to its predecessors, it also has the benefit of improved compatibility with UV encapsulants.
LOCTITE® ABLESTIK ABP 2035SCR cures in low temperatures with high adhesion and excellent dispensing performance. It is ideal for ASIC attach and Lid/Cap attach of optical sensors and as a received die attach for 3d sensing modules. This material is not expected to pass very harsh reliability requirements.
LOCTITE® ABLESTIK ABP 2035SCR has been developed without DHCP. Though it is not banned for use, DHCP has been included on the Substances of Very High Concern (SVHC) list and could eventually be restricted for supply into Europe following future REACH and ECHA regulations. As is listed on the SDS of its predecessors LOCTITE ABLESTIK 2033SC and LOCTITE ABLESTIK 2035SC, DHCP is used as part of the formulation and therefore do contain substances on the SVHC list. So though there is no defined end date for use of ABP 2033SC or ABP 2035SC, when the DCHP substance in both would change from current SVHC list to Annex XIV list, there could be a 3-4 years sunset period on these older products. The "R" in the name thus stands for "REACH future proofing".
Cure Schedule
- 2 minutes @ 120°C
- 30 minutes @ 100°C
- 60 minutes @ 80°C
Technical Specifications
| General Properties | |||||||||
| Work life @25°C Work life @25°C Work life is the amount of time we have to work with a material until it is no longer able to be easily worked and applied on a substrate. It is based on the change in viscosity and it can rely on the application requirements. | 24 hours | ||||||||
| |||||||||
| |||||||||
| Thermal Properties | |||||||||
| Glass Transition Temperature (Tg) Glass Transition Temperature (Tg) The glass transition temperature for organic adhesives is a temperature region where the polymers change from glassy and brittle to soft and rubbery. Increasing the temperature further continues the softening process as the viscosity drops too. Temperatures between the glass transition temperature and below the decomposition point of the adhesive are the best region for bonding. The glass-transition temperature Tg of a material characterizes the range of temperatures over which this glass transition occurs. | 118 °C | ||||||||
| Thermal Conductivity Thermal Conductivity Thermal conductivity describes the ability of a material to conduct heat. It is required by power packages in order to dissipate heat and maintain stable electrical performance. Thermal conductivity units are [W/(m K)] in the SI system and [Btu/(hr ft °F)] in the Imperial system. | 1 W/m.K | ||||||||
| |||||||||
| Mechanical Properties | |||||||||
| |||||||||
| Chemical Properties | |||||||||
| |||||||||
| Physical Properties | |||||||||
| Thixotropic index Thixotropic index Thixotropic Index is a ratio of a material s viscosity at two different speeds in Ambient temperature, generally different by a factor of ten. A thixotropic material s viscosity will decrease as agitation or pressure is increased. It indicates the capability of a material to hold its shape. Mayonnaise is a great example of this. It holds its shape very well, but when a shear stress is applied, the material easily spreads. It helps in choosing a material in accordance to the application, dispense method and viscosity of a material. | 4 | ||||||||
| Viscosity Viscosity Viscosity is a measurement of a fluid’s resistance to flow. Viscosity is commonly measured in centiPoise (cP). One cP is defined as the viscosity of water and all other viscosities are derived from this base. MPa is another common unit with a 1:1 conversion to cP. A product like honey would have a much higher viscosity -around 10,000 cPs- compared to water. As a result, honey would flow much slower out of a tipped glass than water would. The viscosity of a material can be decreased with an increase in temperature in order to better suit an application | 9,830 mPa.s | ||||||||
Additional Information
REACH and RohS FAQ
How can a product be unlisted for several years and then one day it becomes a concern without any composition changes?
We have set REACH regulation compliancy as very high priority. It can happen that MSDS reporting changes to be more strict in line with Regulations such as (EC) No 1907/2006 over time (and will continue to be) . Some DCHP substances are already listed in our MSDS for several years but they can appear above threshold with a simple regulatory change.
What testing and reporting methodology do you apply ?
Any DCHP substances are part of the uncured "as supplied" formulation and reported on MSDS as such. We also submit samples to SGS laboratories for additional reports.
How do you confirm that the other phthalates are under the threshold, even for ROHS ?
With third party RoHS reports showing all RoHS relevant substances with their measurements.
How do you manage the regulatory monitoring about Reach and ROHS ?
Our Product Safety and Regulation Affairs department is monitoring this and update MSDS’s where needed. Next to this, we do third party testing at SGS for volume runners based on specific customer requests.
Would you be able to anticipate the next regulation updates in order to propose your changes in advance ?
We’re following REACH and ECHA regulations closely. If a DCHP substance for instance would change from the SVHC list to Annex XIV list with sunset date 3-4 years ahead, MSDS will be updated and customers will be informed to consider alternative options.
What are the differences between 2033SC and ABP 2035SCR since it contains less DCHP ?
Please check the TDS’s of both 2033SC and ABP 2035SCR next to the slide on ABP 2035SCR below highlighting the benefits. Several other "smart card module makers" already changed from 2033SC to ABP 2035SCR and others are considering this. We are not aware of any negative aspects on ABP 2035SCR vs 2033SC so far.