European REACH Regulation (EC1907/2006) Overview
In short, the goal of the European REACH regulation is to have a clear picture of the types and quantities of chemical suybstances entering the European Union.
On June 1st, 2007, the European REACH (an abbreviation of Registration, Evaluation, Authorisation and Restriction of Chemicals) came into force in the European Union. REACH regulation compliance requires all companies manufacturing or importing chemical substances in the EU to register these substances with the European Chemicals Agency (ECHA).
Does REACH only affect foreign suppliers?
No. REACH affects all suppliers of chemical substances - both doemstic and foreign. With the development and manufacture of so many new substances, ECHA, representing the European Union is trying to get a grasp on what all these new chemical substances are and to understand the risks of using these substances.

European REACH Regulation (EC1907/2006) Services Provider
A complete offering to make you REACH regulation compliant without the hassle
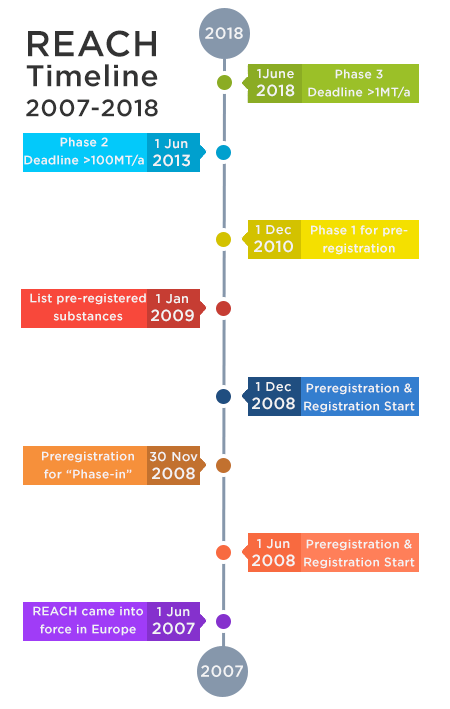
REACH Regulation Timeline
The ECHA recognize that completely understanding the types and quantities of chemical substances into Europe is an enormous undertaking. As is common with such undertakings, the final goal was broken into more manageable deadlines, aiming to understand the largest volume and most critical substances first. An important driver of the deadlines are the volumes of the substances involved. Knowing that new substances will likely begin with lower annual volumes, the first target is the high volume of existing substances.
Below is the timeline that the ECHA has defined for the deadline of specific actions:
- 1 June 2007
REACH regulation came in to force in Europe - 1 June 2008
Pre-registration starts for existing substances. These substances are also called "Phase-in" substances.
Registration starts for new substances. These are also called "Non Phase-in" substances. - 30 November 2008
Pre-registration ends for existing (phase-in) substances - 1 December 2008
Registration starts for existing substances. This excludes substances that have already been pre-registered. - 1 January 2009
List of pre-registered substances was published.The Substance Information Exchange Fora (SIEF) was formed. - 1 December 2010: Phase 1 - The first deadline
This is the date by which the following pre-registered 'phase-in' substances should have been registered when supplied at:
● more than 1000 tonnes per annum (tpa) or;
● more than 100 tonnes per annum and classified under CHIP as very toxic to aquatic organisms or;
● more than 1 ton per annum and classified under CHIP as Cat 1 or 2 carcinogens, mutagens or reproductive toxicants - 1 June 2013: Phase 2 - The second deadline
This is the date for registration of substances supplied at more than 100 tonnes per annum - 1 June 2018: Phase 3 - The third deadline
This is the date for registration of substances supplied at more than 1 ton per annum
REACH Regulation Frequently Asked Questions
What does REACH stand for?
REACH is an abbreviation for the Registration, Evaluation, Authorisation and Restriction of CHemicals and is a European regulation whose goal is to have a clear picture of the types and quantities of chemical substances that are manufactured or imported into the European Union.
Does REACH concern me?
REACH concerns all manufacturers of chemical substances who which to import or sell these substances to European customers. This includes foreign manufacturers, but it also affects European manufacturers who wish to sell to their European customers.
What's the difference between REACH Compliant and ROHS Compliant
REACH compliance has to do with the foreign company. Either your company complies with REACH regulations or you don't. ROHS compliance refers to your products, and specifically checks to see they don't contain six different hazardous materials.
All the labs are full, and I won't get my testing done on time to meet the REACH deadlines. What now?
Anyone rushing to meet the June 1st, 2018 REACH deadline, will recognize that European labs are fully loaded . At a recent REACH ORO meeting, we have recognized that ECHA does allow EU manufacturers and OR's of Non-EU manufacturers to exceed the June 1st registration deadline if they can proof that their delay is caused by a lack of lab time slots.
Furthermore, CAPLINQ has special contracts with labs in the USA and Asia also to help ease some of the current REACH testing burden. Ourr current leatimes for similar services are btween 2-4 weeks.
When does REACH come into effect?
REACH preregistration came into effect on June 1, 2007, but the more important date is January 1, 2018 when REACH registrations are due.
Is the REACH regulation meant to keep out foreign competition?
Absolutely not. It is specifically mentioned in the REACH regulation that REACH is not meant to keep out foreign competition. In fact, as a result of REACH, there will be many hundreds of European manufacturers that will not be REACH compliant either, and so there will be a distinct competitive advantage to those companies who are prepared when REACH is in full force in 2018. CAPLINQ can help give you that competitive edge.
What is the REACH SVHC list?
The REACH regulation aims to understand the potential negative effect of chemical substances on human health and the environment. As such, the ECHA publishes the "Substances of Very High Concern (SVHC)" List as a list of chemicals that are know to either be carcinogens, mutagens, reproductive toxins or persistent, bio-accumulative and toxic chemicals.


